Draw The Lewis Structure Of Co2
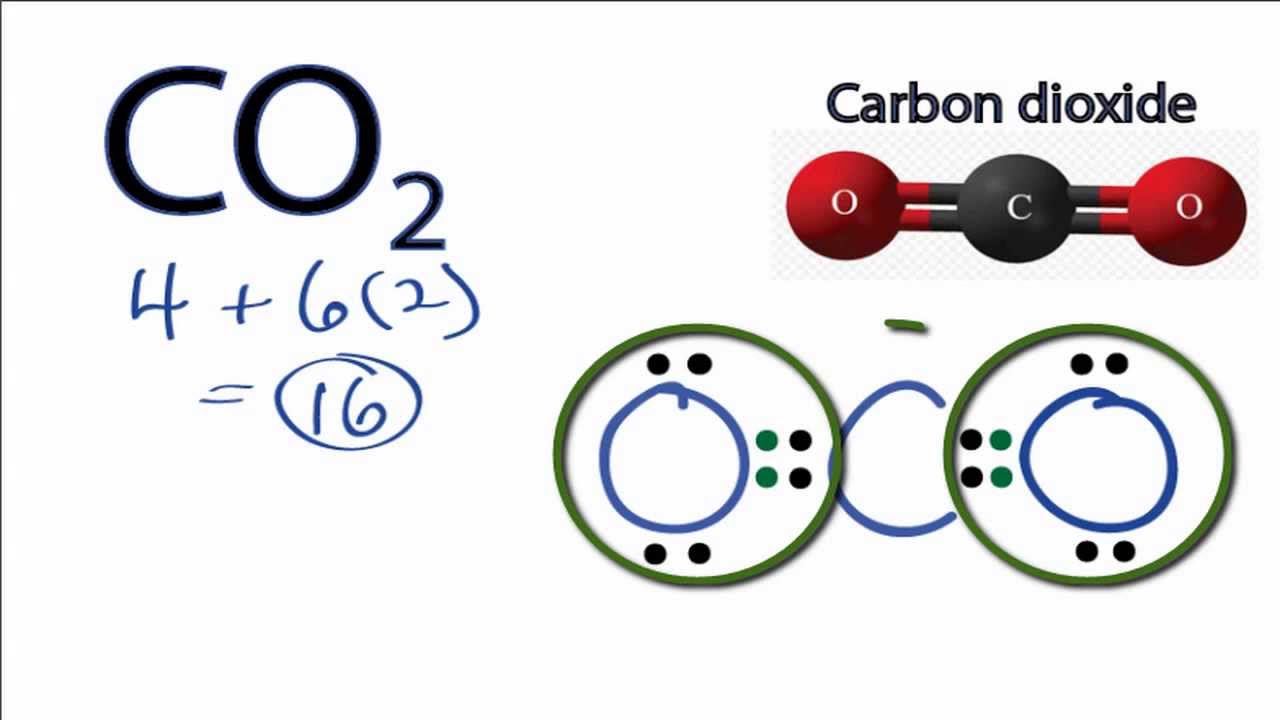
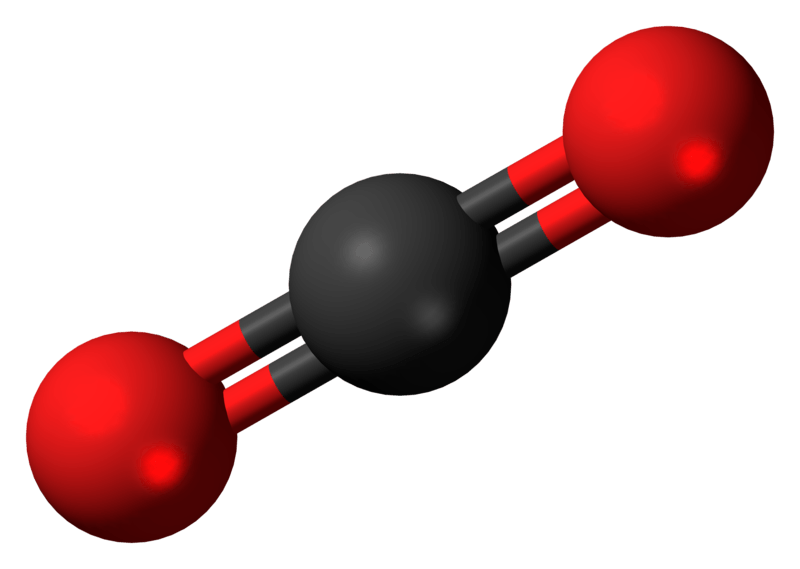
Draw The Lewis Structure Of Co2 - Calculate the total number of valence electrons. In this article, we will explore the lewis structure, molecular geometry,. Calculate the number of valence electrons: On the periodic table, carbon is in group 4, or 14 sometimes; In order to draw the lewis structure of co2, first of all you have to find the total number of valence electrons present in the co2 molecule. Web this video outlines how to draw the lewis structure for co2, or carbon dioxide #chem #chemistry #generalchemistry #lewisstructure #nonpolar #science #co2 #ca. Web the co 2 lewis structure depicts the molecular arrangement of carbon dioxide, which is composed of one carbon atom and two oxygen atoms. Still, 12 electrons are remaining. Web 6 steps to draw the lewis structure of co2 step #1: That will normally be the least electronegative atom (c). Web carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. Carbon (c) and oxygen (0). Draw a trial structure by putting. Web learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Web following up the step and after drawing the skeletal diagram, we see that 4 electrons are used so far. It can be liquified and even frozen solid with special machinery to produce “dry ice.” Decide which is the central atom in the structure. Web you follow a sequence of steps. Here, the given molecule is co2 (carbon dioxide). Understanding the structure and properties of co2 is essential for comprehending its behavior and its impact on the environment. Web in this article, we will explore the co2 lewis structure, how to draw it, and its properties and applications in chemistry. Web this chemistry video explains how to draw the lewis structure of co2 also known as carbon dioxide. Web how to draw lewis structure of co2. In this case, we can condense the last few steps, since not. No lone pairs on carbon atom and each oxygen atom has two lone pairs on their valence shells. Decide which is the central atom in the structure. Understanding the structure and properties of co2 is essential for comprehending its behavior and its impact on the environment. Web how to draw lewis structure of co2. Web we can draw the lewis. Generally, small symmetric molecules are nonpolar. Carbon has 4 valence electrons, while each oxygen atom has 6. Web this video outlines how to draw the lewis structure for co2, or carbon dioxide #chem #chemistry #generalchemistry #lewisstructure #nonpolar #science #co2 #ca. Determine the total number of valence electrons. Web carbon needs two double bonds, one to each of the two oxygens,. This molecule can be a good start for beginners who want to learn the fundamentals of such concepts and want to know how to draw lewis dot structures for other molecules as well. Draw a trial structure by putting. Web the co 2 lewis structure is symmetric. The carbon atom is sp hybridized and oxygen atoms are sp2, making the. Web carbon needs two double bonds, one to each of the two oxygens, to complete its octet. In the earth's atomsphere it is considered a greenhouse gas. 8 + (6 × × 7) = 50; In this case, we can condense the last few steps, since not all of them apply. Carbon (c) and oxygen (0). Web this chemistry video explains how to draw the lewis structure of co2 also known as carbon dioxide. Generally, small symmetric molecules are nonpolar. Understanding the structure and properties of co2 is essential for comprehending its behavior and its impact on the environment. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed. On the periodic table, carbon is in group 4, or 14 sometimes; Draw a skeleton joining the atoms by single bonds. So, first, we must determine the valence electrons of these two atoms independently. Web carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. Two double bonds connect the carbon and oxygen atoms in the. We're going to do the lewis structure for co2, carbon dioxide. Web carbon dioxide (co2) lewis structure has two double bonds around carbon atom. Still, 12 electrons are remaining. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Web this video outlines how to. > here are the steps that i follow when drawing a lewis structure. Calculate the number of valence electrons: Determine the total number of valence electrons. Generally, small symmetric molecules are nonpolar. Within the co 2 lewis structure, the carbon atom is surrounded by two double bonds, with each oxygen atom attached to it. The carbon atom is sp hybridized and oxygen atoms are sp2, making the overall. Draw a trial structure by putting. Each oxygen atom possesses two lone pairs, while the carbon atom. In the earth's atomsphere it is considered a greenhouse gas. Steps of drawing the lewis structure of co2 are explained. In order to draw the lewis structure of co2, first of all you have to find the total number of valence electrons present in the co2 molecule. Web you follow a sequence of steps. Web carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. 8 + (2 × × 7) = 22 xef 6: I also go over hybridization, shape and bond angles. Draw a skeleton joining the atoms by single bonds. Web this video outlines how to draw the lewis structure for co2, or carbon dioxide #chem #chemistry #generalchemistry #lewisstructure #nonpolar #science #co2 #ca. Shape of co2 is linear. So, first, we must determine the valence electrons of these two atoms independently. Web to draw the co2 lewis structure we have to follow a few steps. It can be liquified and even frozen solid with special machinery to produce “dry ice.” Web carbon dioxide (co2) is a compound that plays a crucial role in various fields including chemistry, biology, and environmental science. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Carbon has 4 valence electrons, while each oxygen atom has 6 valence electrons. Web carbon dioxide (co2) lewis structure has two double bonds around carbon atom. Generally, small symmetric molecules are nonpolar.CO2 Lewis Structure How to Draw or Write the Lewis Dot Structure for
CO2 (Carbon Dioxide) Lewis Dot Structure Science Trends
What is the Lewis Dot structure for CO2 (Carbon dioxide)?
Carbon Dioxide Lewis Structure How to Draw the Lewis Structure for
Lewis Structure of CO2 [with video and free study guide]
[Solved] draw the lewis structure of CO2. include lone pairs Draw the
CO2 (Carbon Dioxide) Lewis Dot Structure Science Trends
Complete Guide To Drawing Lewis Dot Structures and Formal Charges
CO2 Lewis Structure How to Draw the Dot Structure for Carbon Dioxide
Lewis Structure Definition and Example
Carbon (C) And Oxygen (0).
Web To Draw The Lewis Structure Of An Atom, Write The Symbol Of The Atom And Draw Dots Around It To Represent The Valence Electrons.
Draw A Trial Structure By Putting.
This Molecule Can Be A Good Start For Beginners Who Want To Learn The Fundamentals Of Such Concepts And Want To Know How To Draw Lewis Dot Structures For Other Molecules As Well.
Related Post:



![Lewis Structure of CO2 [with video and free study guide]](https://www.aceorganicchem.com/blog/wp-content/uploads/2023/05/CO2-lewis-puzzle.jpg)

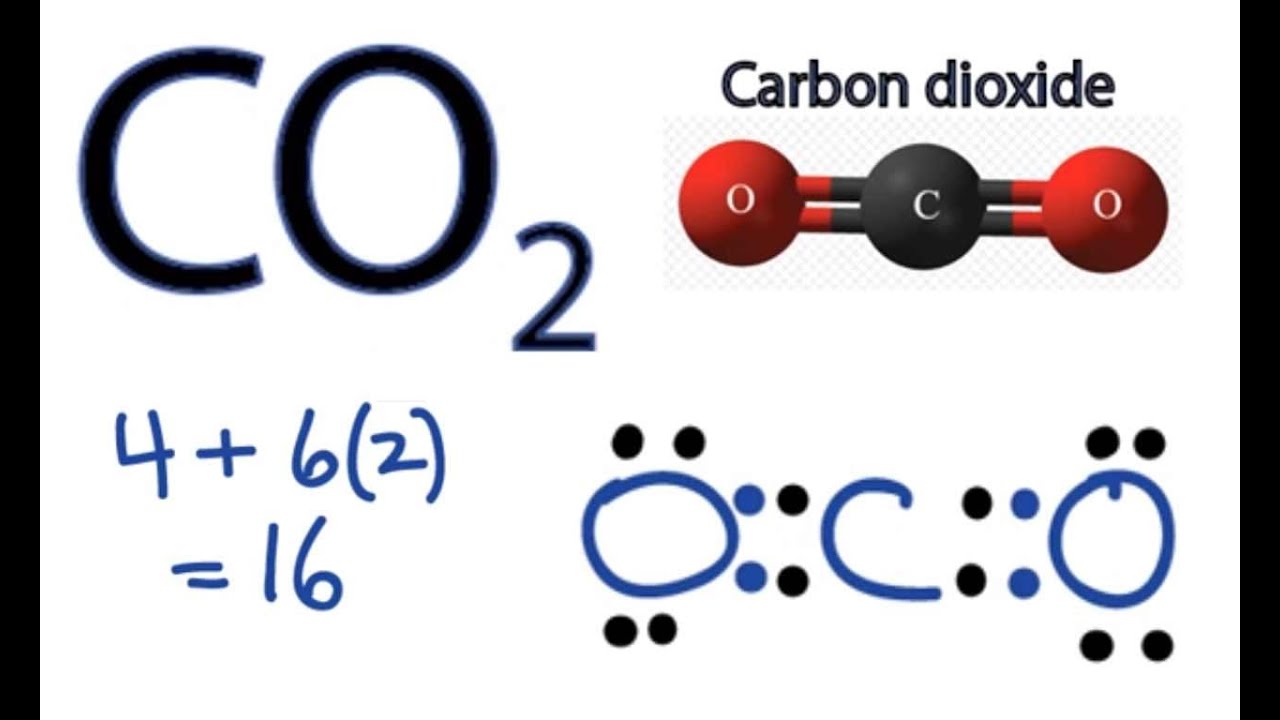
:max_bytes(150000):strip_icc()/CO2LewisStructure-591c94063df78cf5fadfde77.png)